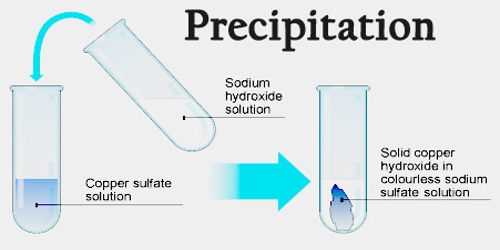
There are also some general rules you can learn for the solubility of different compounds. A great resource is finding a good solubility table or solubility chart. These are general guidelines or rules on what compounds will form a precipitate. Both the charge and the number of atoms of each element must be balanced. Net ionic equations must be balanced to be accurate. What is left is called the net ionic equation. These ions are also called spectator ions. To simplify this equation further, get rid of any ion that appears on both the reactant and product side as that indicates they are not part of the reaction. This format is called the complete ionic equation:Īg +(aq) + NO 3 –(aq) + Na +(aq) + Cl –(aq) –> AgCl(s) + Na +(aq) + NO 3 –(aq) So the reaction including all components would be:ĪC(aq) + BD(aq) –> AD(s) + B +(aq) + C –(aq)ĪgNO 3(aq) + NaCl (aq) –> AgCl(s) + Na +(aq) + NO 3 –(aq)īecause the reactants are aqueous and we want to know the ions in solution, it is common to write the reaction in terms of the ions. The reactants must be ionic compounds in solution. The most general form of a precipitation reaction then is: The product must include a solid product. The reactants are usually two or more ionic aqueous molecules. How to Identify a Precipitation ReactionĪ precipitation reaction will always have a solid product. These reactions are commonly used to help determine what ions are in the solution. The liquid left behind is referred to as the supernatant. The solid particles can then also be removed from the solution by various means such as filtration, decantation, centrifuging. In precipitation reactions, the formed precipitate can remain suspended in solution or may sink to the bottom. AB and CD are both ionic compounds aqueous in solution. To be a precipitate reaction, either AD or CB will be an insoluble solid. This type of reaction takes the following form. The other pair of cation and anion may or may not be soluble. That is when there are two salts that are soluble, and when the cation of one binds with the anion of the other it forms a solid product that is not soluble. Often, a precipitation reaction is a ‘double replacement reaction’. Once the precipitate has been recovered, the resulting powder may be called a "flower.Lead iodide precipitating out of solution as a solid compound (from Wikipedia Commons) A common sedimentation technique is centrifugation. Sedimentation refers to any procedure that separates the precipitate from the liquid portion of the solution, which is called the supernate. 
If the particle size of the insoluble compound is very small or there is insufficient gravity to draw the solid to the bottom of the container, the precipitate may be evenly distributed throughout the liquid, forming a suspension. The solid that is formed is called the precipitate. 
A chemical that causes a solid to form in a liquid solution is called a precipitant. Here's how it works: forming a solid from a solution is called precipitation. The terminology can seem a bit confusing. They are used for purification, removing or recovering salts, for making pigments, and to identify substances in qualitative analysis. Precipitation reactions serve important functions.The solid that forms via a precipitation reaction is called the precipitate.To precipitate is to form an insoluble compound, either by decreasing the solubility of a compound or by reacting two salt solutions. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
